These structures and chemical ingredients though various result in universally recognized ceramic like properties of enduring utility including the following.
Key chemical properties of ceramics.
Low electrical and thermal conductivity they re good insulators.
Great hardness and strength.
Basically these bonds result in good chemical resistance but have the low thermal expansion high melting point and hardness.
Ceramics exhibit very strong ionic and or covalent bonding stronger than the metallic bond and this confers the properties commonly associated with ceramics.
Chemical inertness they re unreactive with other chemicals.
Ceramics are hard and strong but brittle.
Hardness contributing to resistance against wear.
Composite materials combine two or more materials.
Ceramic tiles are highly resistant to harsh chemical agents like alkalis acids household chemicals and swimming pools salts in high and low concentrations.
High melting points so they re heat resistant.
Advanced ceramics fine ceramics possess good chemical stability.
And an ability to take a decorative finish.
Ceramics are hard and strong but brittle.
Different materials have different properties.
Composite materials combine two or more materials.
Polymers are strong and tough and often flexible.
High hardness high compressive strength low thermal and electrical conductivity and chemical inertness.
Thermal and electrical conductivity considerably lower than that of metals.
Melting points high 600 4000c thermal conductivities are low insulators thermal expansion values are low 1 15 ppm c 3.
Low to medium thermal conductivity.
A ceramic material is an inorganic non metallic often crystalline oxide nitride or carbide material.
Different materials have different properties.
Some elements such as carbon or silicon may be considered ceramics.
If we re summarizing their properties we can say that ceramics have.
Considerable durability they re long lasting and hard wearing.
Low to medium tensile strength.
The properties of ceramics make fracturing an important inspection method.
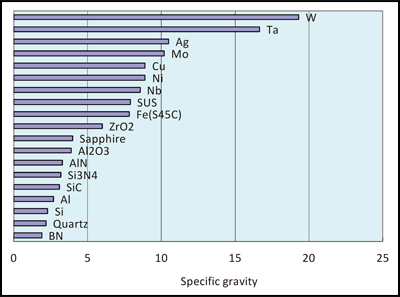
Mass properties e g density ceramics are intermediate density 2 00 6 00 gms cm3 different for allotropes e g glass cristobalite tridymite quartz 2.
Mechanical strength in spite of brittleness.
Ceramics are bonded together by an ionic or covalent bond.
High resistance to corrosion and chemical attack.
Chemical durability against the deteriorating effects of oxygen water acids bases salts and organic solvents.